


For decades, creatine monohydrate (CM) has been associated with bodybuilding and elite athletics – a limiting perspective increasingly incompatible with modern data. As healthcare systems transition toward value-based care, clinicians are being asked to prioritize interventions that are clinically effective, safe across populations, and economically efficient. Within this framework, the American Academy of Value-Based Care (AAVBC) spotlights therapies that improve longitudinal outcomes while reducing downstream utilization.1

A robust body of literature now identifies creatine as a fundamental metabolic regulator with multiple mechanisms which benefits cardiovascular health, neuroprotection, and the management of age-related physiological decline.2 Creatine (N-(aminoiminomethyl) -N-methyl glycine) is a naturally occurring nitrogenous compound essential to cellular energy homeostasis.2 More than 1,000 peer-reviewed publications support its safety and efficacy across athletic and non-athletic populations, including older adults and patients with chronic disease.2 This expanding evidence base demands us to reconsider creatine as a metabolic support therapy with clinically meaningful functional effects, not merely a performance supplement.
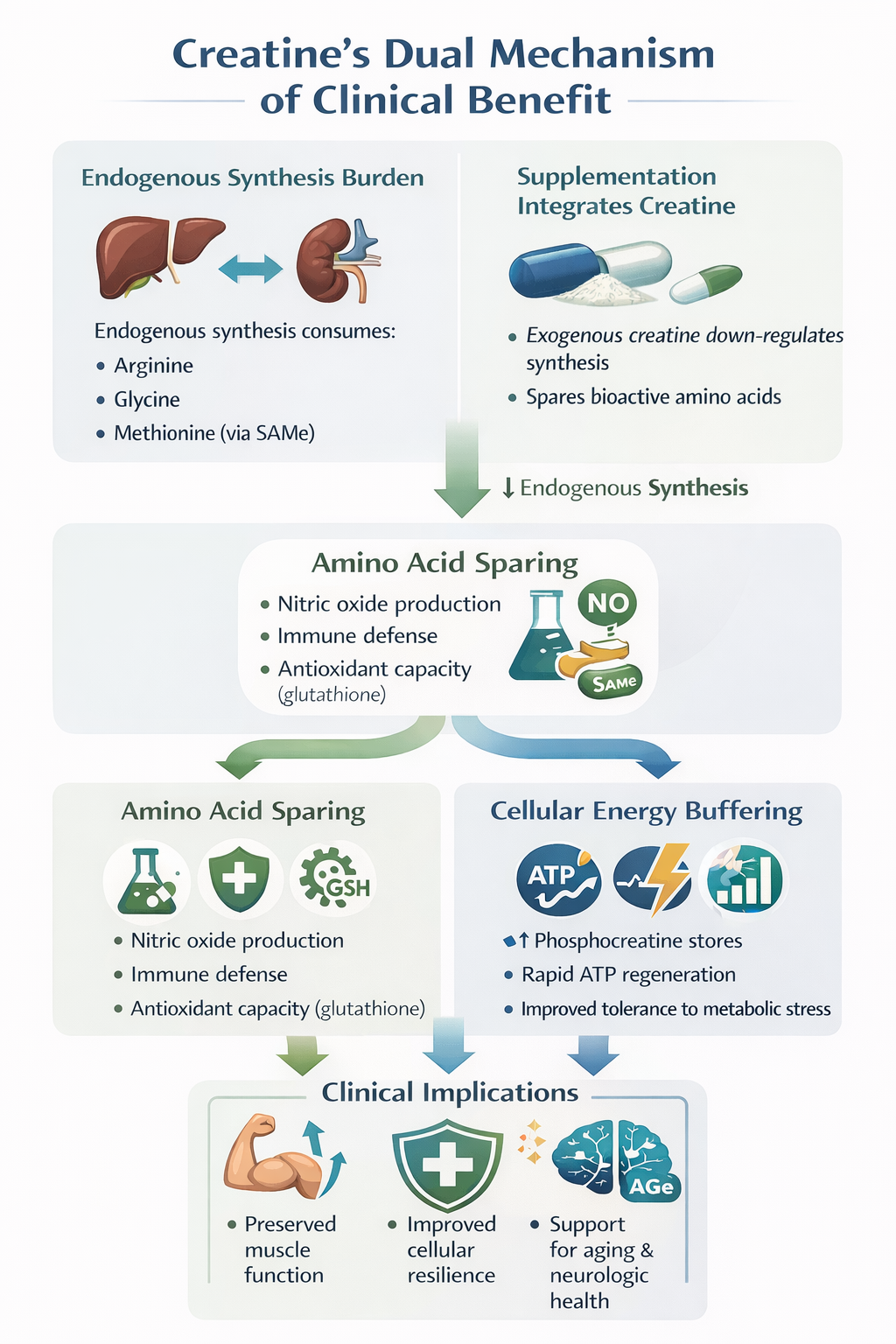
Endogenous creatine synthesis occurs via a two-step inter-organ pathway involving the kidneys and liver.3 The enzyme L-arginine: glycine amidinotransferase (AGAT) converts arginine and glycine to guanidinoacetate (GAA), which is subsequently methylated by guanidinoacetate N-methyltransferase (GAMT) using S-adenosylmethionine (SAMe) as a methyl donor.3 This process imposes a significant metabolic burden.
In a typical 70-kg adult, endogenous creatine synthesis (~1.5–2 g/day) consumes approximately 46% of daily arginine intake, 36% of glycine, and up to 87% of methionine requirements.4 By providing exogenous creatine monohydrate through supplementation, the body can downregulate this production, effectively "sparing" these bioactive amino acids for other critical functions.3,4
This sparing effect has meaningful clinical implications. Preserved arginine availability supports nitric oxide (NO) production and immune defense (NO is a killer of pathogenic bacteria, fungi, parasites, and viruses), while methionine conservation maintains SAMe pools required for DNA methylation and glutathione synthesis, the body’s principal antioxidant system.4,5 From a value-based care perspective, this represents a rare example of a single low-cost intervention improving multiple metabolic pathways simultaneously.
Creatine’s primary biological role is as a high-energy phosphate buffer in tissues with fluctuating energy demands, including skeletal muscle, myocardium, and the brain.6 After cellular uptake via the SLC6A8 creatine transporter, creatine is phosphorylated by creatine kinase to phosphocreatine (PCr).6
During periods of metabolic stress, PCr rapidly donates phosphate groups to regenerate adenosine triphosphate (ATP):
PCr + ADP (low energy) + H⁺ ⇌ ATP (high energy) + Cr
This system functions as both a temporal buffer, sustaining ATP during sudden energy demand, and a spatial buffer, shuttling high-energy phosphates from mitochondria to cytosolic sites of utilization.6 Supplementation increases intramuscular and cerebral PCr stores by approximately 10 - 40%, improving tolerance to metabolic stress without altering resting ATP levels.2
Aging is associated with a progressive decline in creatine and PCr availability, particularly in skeletal muscle.7 Research has shown that Phosphocreatine regeneration rates decrease by approximately 8% per decade after age 30. This reduction is closely associated with the atrophy of Type II (fast-twitch) muscle fibers, which are highly dependent on the phosphagen system for power.6,7
Randomized trials demonstrate that creatine supplementation combined with resistance training produces significantly greater gains in lean mass, strength, and functional performance in older adults compared with resistance training alone.8 These improvements are clinically relevant: lower-body strength is a key determinant of fall risk, fracture incidence, and subsequent hospitalization in aging populations.7, 8 For value-based organizations accountable for total cost of care, preserving musculoskeletal function directly supports utilization reduction.
When initiating any supplementation, clinicians should balance anticipated clinical benefits against potential risks, individualized to each patient’s comorbidities, renal function, medication profile, and overall health goals. Even supplements with extensive safety data are not universally appropriate. Variability in baseline health status, dosing practices, product quality, and monitoring can influence outcomes. A prudent approach includes reviewing existing laboratory markers, assessing contraindications, and educating patients about evidence-based dosing rather than assuming safety solely on the basis of widespread use.
Concerns regarding kidney injury stem largely from misinterpretation of serum creatinine levels. Creatinine is a breakdown product of creatine; supplementation may raise serum creatinine without impairing glomerular filtration rate or causing renal pathology.2,9 Long-term studies using doses up to 30 g/day for up to five years demonstrate that creatine is safe in individuals with normal renal function.2,9
Caution: It is paramount to weigh possible clinical benefits with pre-existing risk factors. For those with pre-existing kidney disease or risk factors like diabetes and hypertension, caution is advised as long-term data in these populations are limited. In these cases, clinicians may prefer to monitor Cystatin C, a biomarker of kidney function that is unaffected by creatine intake. Overall, it is recommended to avoid supplementation when clear risks are present; creatine should likewise be avoided, as the benefits are likely to outweigh the risks.
Creatine increases intracellular water content within muscle cells to maintain osmotic balance.2 This phenomenon reflects cellular hydration rather than pathologic fluid retention and may act as an anabolic signal promoting protein synthesis.7 Maintenance dosing (3–5 g/day) does not result in clinically meaningful long-term increases in total body water relative to lean mass.2,10
Speculation regarding hair loss originates from a single small study reporting increased dihydrotestosterone (DHT) levels that remained within normal clinical ranges and did not measure alopecia.9 Subsequent studies have failed to replicate this finding.9
The brain accounts for roughly 20% of total energy consumption despite representing only ~2% of body weight.11 Creatine enhances cerebral energy buffering and reduces vulnerability to ischemia, oxidative stress, and mitochondrial dysfunction.6,11 Although brain uptake is slower than in skeletal muscle, higher or prolonged dosing (10–20 g/day) can meaningfully increase brain creatine stores.11
Emerging evidence supports creatine’s potential beneficial role in:
While not a disease-modifying therapy, creatine represents a biologically plausible neuroprotective adjunct.
Creatine’s medical legitimacy is most clearly demonstrated in gyrate atrophy, a rare inherited disorder (roughly 1 in 1,000,000 people globally) caused by ornithine aminotransferase deficiency.25 Excess ornithine inhibits AGAT, leading to systemic creatine deficiency and progressive muscle atrophy.25,26 In this context, creatine monohydrate functions as a necessary medical food, restoring muscle PCr levels and improving functional outcomes.26,27
Evidence-based protocols are straightforward:
Creatine monohydrate remains the gold standard; alternative formulations offer no consistent clinical advantage.2
Creatine monohydrate exemplifies the type of intervention value-based healthcare should prioritize: low cost, broad physiologic relevance, exceptional safety, and measurable functional benefit. Far from being limited to athletic performance, creatine supports musculoskeletal integrity, cerebral bioenergetics, immune metabolism, and healthy aging.2,6,7
The evidence no longer supports relegating creatine to the supplement aisle. In modern clinical practice, it represents a rational, evidence-aligned metabolic therapy that fits squarely within value-based care principles.
The above content is for informational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health providers with any questions about a medical condition.